The Gap Analysis is one of the most important steps in your project. It outlines what areas of your current Laboratory System needs to migrate from ISO 9001:2015 to ISO 17025:2017.
This gap analysis checklist is prepared to highlight the differences of ISO 17025:2017 and ISO 9001:2015.
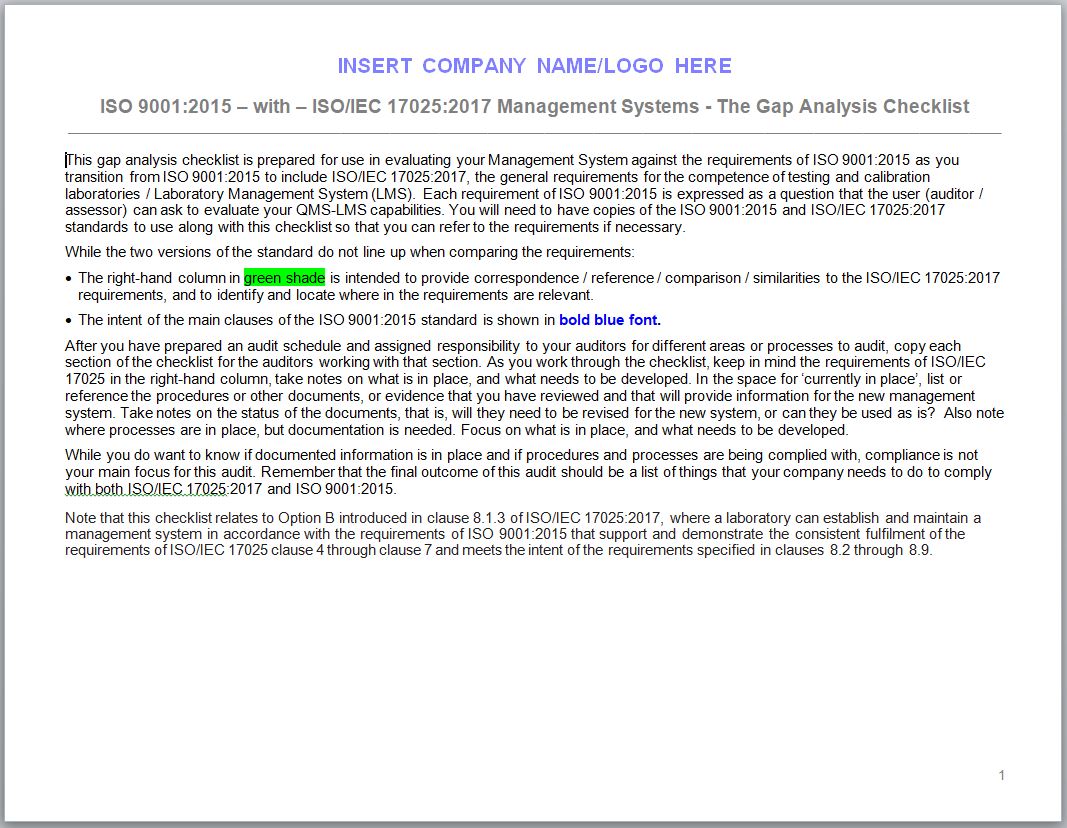
This gap analysis checklist is prepared for use in evaluating your Management System against the requirements of ISO 9001:2015 as you transition from ISO 9001:2015 to include ISO/IEC 17025:2017, the general requirements for the competence of testing and calibration laboratories / Laboratory Management System (LMS).
Each requirement of ISO 9001:2015 is expressed as a question that the user (auditor / assessor) can ask to evaluate your QMS-LMS capabilities. You will need to have copies of the ISO 9001:2015 and ISO 17025:2017 standards to use along with this checklist so that you can refer to the requirements if necessary.
While the two versions of the standard do not line up when comparing the requirements:
- The right-hand column in green shade is intended to provide correspondence / reference / comparison / similarities to the ISO 17025:2017 requirements, and to identify and locate where in the requirements are relevant.
- The intent of the main clauses of the ISO 9001:2015 standard is shown in bold blue font.
- Includes Technical Support
- 70 page Gap Analysis Checklist
Compare Products
Which version is right for me?
If you are transitioning from ISO 17025:2005 to ISO 17025:2017, the Gap Transition Checklist provides you a map of where to copy some text from your existing LMS, and place it in a new LMS structure.
If you are adding ISO 17025:2017 to ISO 9001:2015 structure, the ISO 9001:2015 to ISO 17025:2017 Gap Checklist highlights the differences with ISO 9001:2015 requirements and ISO 17025:2017.