They are written in Microsoft Word and Excel format and are ready to customize for your organization.
These forms have been designed to be integrated with our Laboratory Quality Manual and Procedures. Save by purchasing the Laboratory Quality Manual, Procedures and Forms package.
Complete set of ISO 17025:2017 Forms
| F-510-001 | LMS-Process identification worksheet |
| F-570-001 | Comment and suggestion report |
| F-610-001 | Equipment problem report |
| F-610-002 | Resource maintenance record |
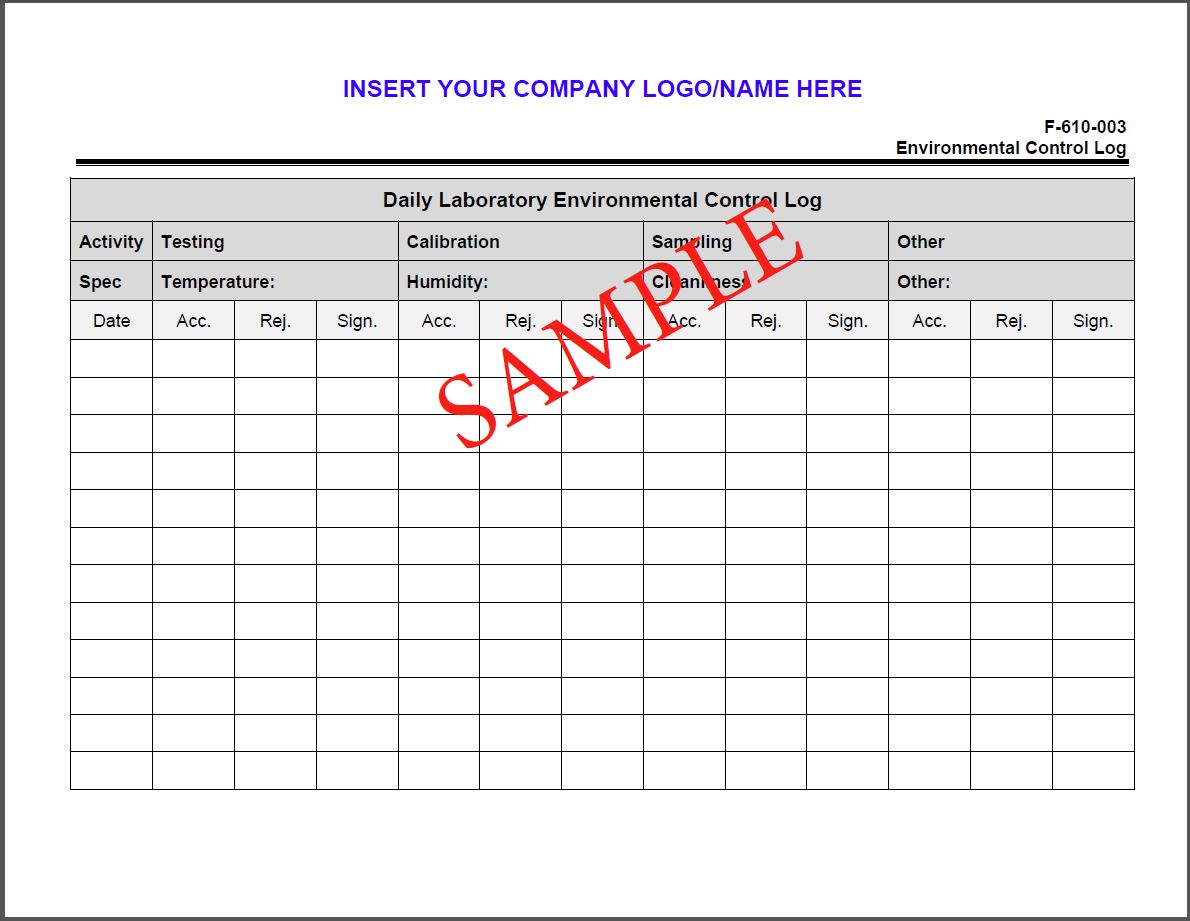
| F-610-003 | Environmental control log |
| F-620-001 | Training action plan |
| F-620-002 | Group training record |
| F-620-003 | Job description |
| F-620-004 | Employee training summary |
| F-645-001 | Equipment calibration list |
| F-660-001 | Provider assessment report |
| F-660-002 | List of acceptable sources |
| F-660-003 | Provider corrective action request – PCAR |
| F-660-004 | Purchase requisition |
| F-660-005 | Purchase order |
| F-660-006 | Business agreement – contract |
| F-710-001 | Client assessment report |
| F-710-002 | Order notification |
| F-720-001 | Project planning worksheet |
| F-720-002 | Method routing summary |
| F-740-001 | Storage inspection report |
| F-740-002 | External property control log |
| F-740-003 | Identification tag / label |
| F-740-004 | Laboratory activity log |
| F-755-001 | Monitoring report |
| F-780-001 | Statement of delivery / invoice |
| F-790-001 | Complaint response report |
| F-790-002 | Nonconformance report |
| F-820-001 | List of documented information |
| F-820-002 | Records matrix |
| F-820-003 | Master documentation lists |
| F-820-004 | Quality Records Table |
| F-820-005 | Document change request form |
| F-820-006 | Document revision checklist |
| F-820-007 | Software inventory spreadsheet |
| F-820-008 | Revision status form |
| F-850-001 | LMS-Risk and opportunity worksheet |
| F-850-002 | LMS-Objectives planning record |
| F-860-001 | Data analysis worksheet |
| F-860-002 | Customer survey and analysis |
| F-870-001 | Corrective action request – CAR |
| F-880-001 | Applicable procedure by work area |
| F-880-002 | Internal audit checklist |
| F-880-003 | Audit plan |
| F-880-003 | Audit report |
| F-890-001 | Management review agenda |
| F-890-002 | Management review output report |
Includes Technical Support
Which version is right for me?
If you plan to reconfigure your existing quality manual and procedures to meet the ISO 17025:2017 standard, use Upgrade Instructions to create everything on your own. They will tell you where to make the changes but will NOT provide templates or new verbiage on the new requirements.
ISO 17025:2017 LQMS is for those just creating their first ISO Management System, and provides necessary documentation to meet the requirements of ISO 17025:2017. See the informational box above for samples, contents, and more information.
If you are transitioning from ISO 17025:2005 to ISO 17025:2017, the LQMS Upgrade includes the QMS + instructions. This provides you a map of where to copy some text from your existing QMS, and place it in a new QMS structure which follows Annex L.
Compare Products
The Forms are available in this Forms Package or as part of the Laboratory Quality Manual & Procedures Package: