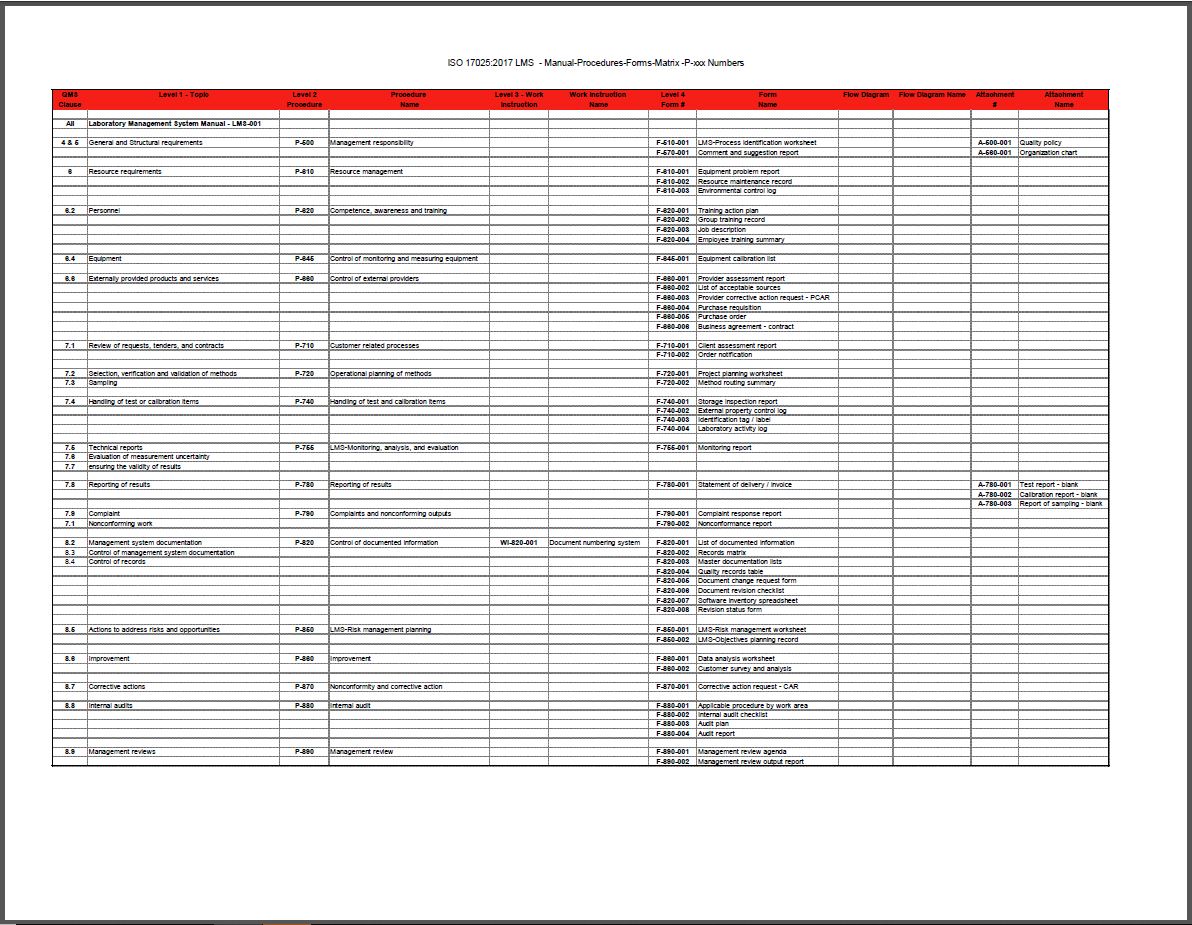
This fully customizable Laboratory Quality Manual and Procedures Package not only includes all the processes and documents you need, but also our ISO/IEC 17025:2005 to 2017 LQMS Upgrade instructions.
As you implement your ISO 17025 system you will also need checklists and training. You can save time and money by purchasing our Certification Packages!
You have the 2005 version in place and now have the objective of upgrading the system to the 2017 version. The good news is that since you are familiar with formal management systems, this initiative will be relatively straightforward.
- The detailed instructions focus on the areas of the documentation required for ISO 17025:2017.
- The intent of the main clauses is shown in blue font,
- The text in italics in the 2nd right hand column summarizes the requirements and indicates where they were included in previous ISO 17025:2005.
- The corresponding clauses for the standards are highlighted in yellow.
- Use a copy of the ISO 17025:2017 standard along with this instruction to pinpoint for your organization the areas that need attention.
- Make notes and add comments in the space available to the right and the left of the column for reference documentation.
- Use the upgrade checklist section on the right side of the table to assign the responsibility for the upgrade and to follow up on its completion.
- ISO/IEC 17025:2005 to 2017 LQMS Upgrade instructions.
- One condensed Manual to introduce the documented information required for ISO 17025:2017.
- A group of procedure/system documents in your LMS with updates to reflect a document numbering system related to the new clause numbers and to incorporate the upgrades for ISO 17025:2017 requirements.
- A group of forms and attachments needed for the documented information and systems.
Compare Products
Which version is right for me?
If you plan to reconfigure your existing quality manual and procedures to meet the ISO 17025:2017 standard, use Upgrade Instructions to create everything on your own. They will tell you where to make the changes but will NOT provide templates or new verbiage on the new requirements.
ISO 17025:2017 LQMS is for those just creating their first ISO Management System, and provides necessary documentation to meet the requirements of ISO 17025:2017. See the informational box above for samples, contents, and more information.
If you are transitioning from ISO 17025:2005 to ISO 17025:2017, the LQMS Upgrade includes the QMS + instructions. This provides you a map of where to copy some text from your existing QMS, and place it in a new QMS structure which follows Annex L.
| Price Each | 17025:2017 QMS |
17025:2005 to 17025:2017 QMS |
|
|---|---|---|---|
| Your Price | |||
| ISO 17025:2017 Laboratory Quality Manual (not sold separately) | $149 | ||
| ISO 17025:2017 Procedures and Work Instructions | $400 (18) | ||
| ISO 17025:2017 Forms and Attachments | $109 (52) | ||
| ISO 17025:2005 to 2017 LQMS Upgrade Instructions | $299 | ||
| Support | FREE | ||
| Your Price |